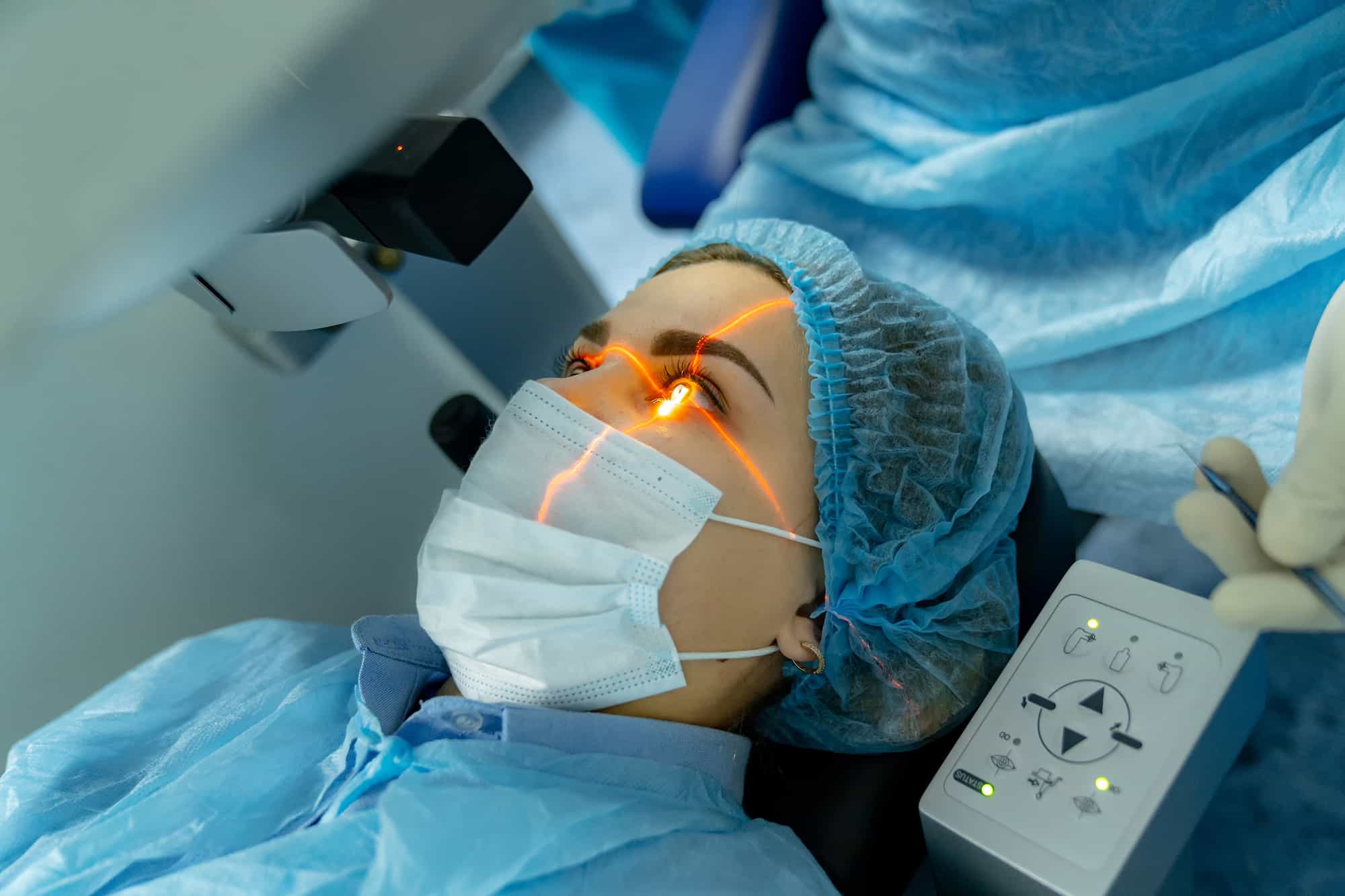
Life Science
Precision Laser Integration for Life Science Systems
Custom, pre-aligned laser assemblies engineered for accuracy, repeatability, and reliability in regulated medical and diagnostic environments.

Differentiators
Engineered for Clinical Confidence
Regulatory-Ready
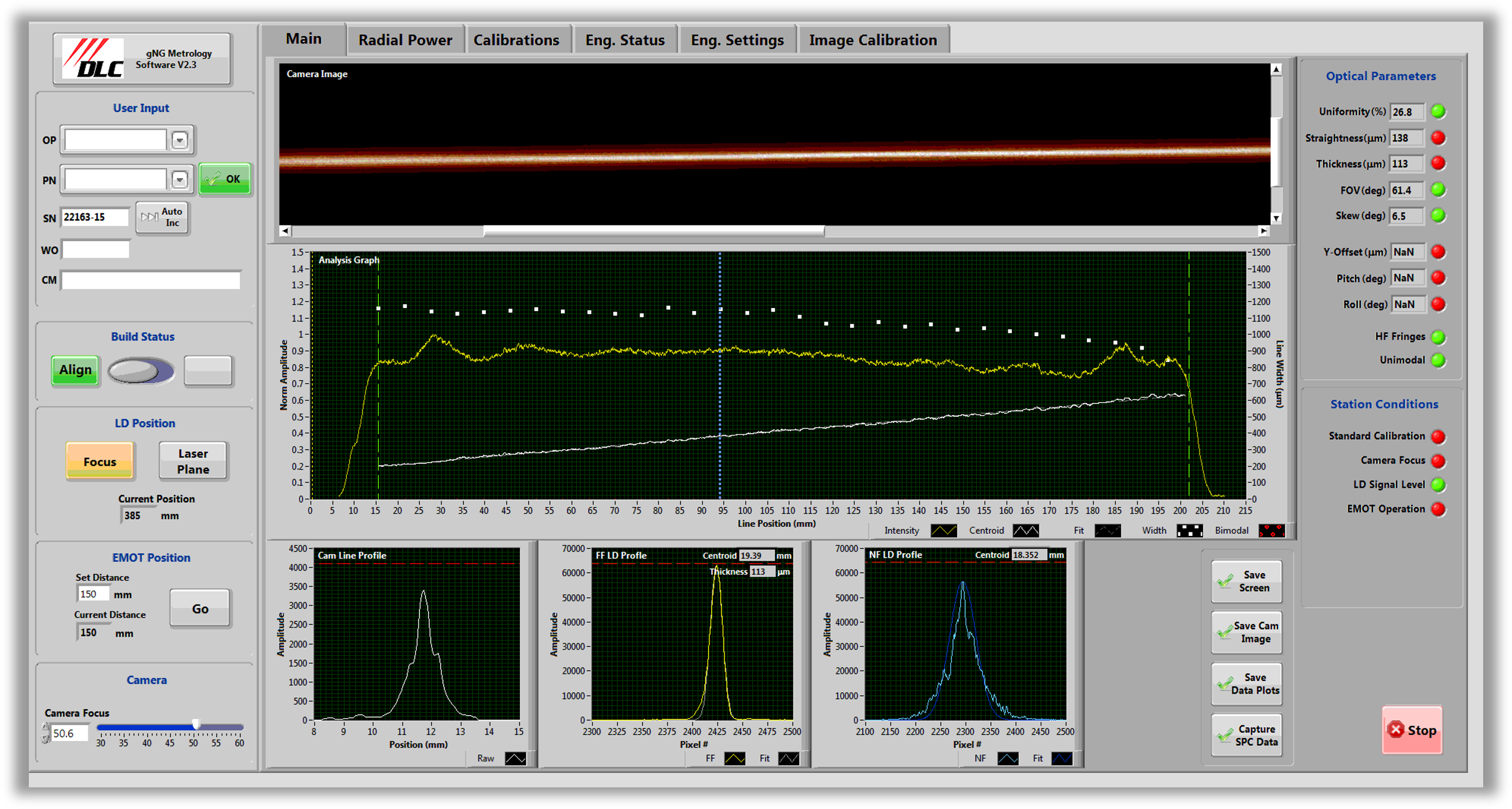
Controlled processes and 100% inspection support FDA-regulated environments.
Pre-Aligned
Pre-aligned laser assemblies ensure accuracy in diagnostic and therapeutic systems.
Pre-Calibrated
Pre-calibration supports precise output control for patient-facing devices.
Traceable
Documented processes and inspection records support audits and compliance.
Scalable
U.S. manufacturing with flexible volume options maintains consistency.
Proven
Designed for long-term reliability in medical and laboratory use.
Applications
Precision Engineered for Medical Devices
DLC supports a wide range of medical and life-science applications where accuracy, stability, and reliability are essential.
- Patient positioning lasers for medical imaging systems
- Cold laser therapies for pain management, fat reduction, and veterinary care
- Laser aiming and alignment for surgical and diagnostic devices
2D and 3D scanning for wound monitoring and imaging - Flow cytometry and fluorescence for clinical diagnostics
- Fluid monitoring and sensing in medical equipment
- Laser-based safety systems for detection and fail-safe shutdown

Case Studies
Featured Case Studies
Does your business need help designing Custom Laser Solutions?

Our Process
Complete Laser Integration, From Design to Manufacturing
DLC combines optical design, mechanical engineering, and in-house manufacturing to deliver turnkey diode laser assemblies built to your exact specifications.
Design
Application-specific laser engineering optimized for performance and integration.
Development
Turning designs into production-ready laser systems through engineering.
Rapid Prototyping
Fast iterative prototypes to validate designs and accelerate decisions.
Advanced Manufacturing
Scalable controlled manufacturing delivering consistent quality at production.
FAQs
Everything You Need to Know
Helpful clarity on our experience, process, and what to expect when working with the DLC team.
Is DLC an ISO 13485 certified medical device manufacturer?
DLC operates under ISO 9001 certified processes. This supports FDA-regulated OEMs by ensuring controlled manufacturing, inspection, and quality consistency.
How do you ensure quality and traceability?
Can you support both prototypes and production volumes?
How do you manage supply-chain reliability for medical components?